SURPASS-CVOT Trial
Landmark phase 3 cardiovascular outcome trial comparing tirzepatide with dulaglutide in type 2 diabetes
The long awaited SURPASS-CVOT trial was published on December 17, 2025, several months after the topline results were presented in September 2025 at the European Association for the Study of Diabetes (EASD) conference in Vienna, Austria!
SURPASS-CVOT. N Engl J Med 2025;393:2409-2420
Bottom line and take home messages
The SURPASS-CVOT trial (2025)1 found that for patients with type 2 diabetes and established atherosclerotic cardiovascular disease, treatment with tirzepatide was noninferior, but not superior, to dulaglutide for reducing major adverse cardiovascular events over a median follow‑up of approximately 4 years.
Tirzepatide was noninferior to dulaglutide for the primary composite endpoint of cardiovascular death, MI, or stroke, with events occurring in 12% vs 13% of participants (HR 0.92). Additional analyses reported greater reductions in A1C, weight, renal measures, and all‑cause mortality with tirzepatide compared with dulaglutide, though these were not multiplicity‑controlled.
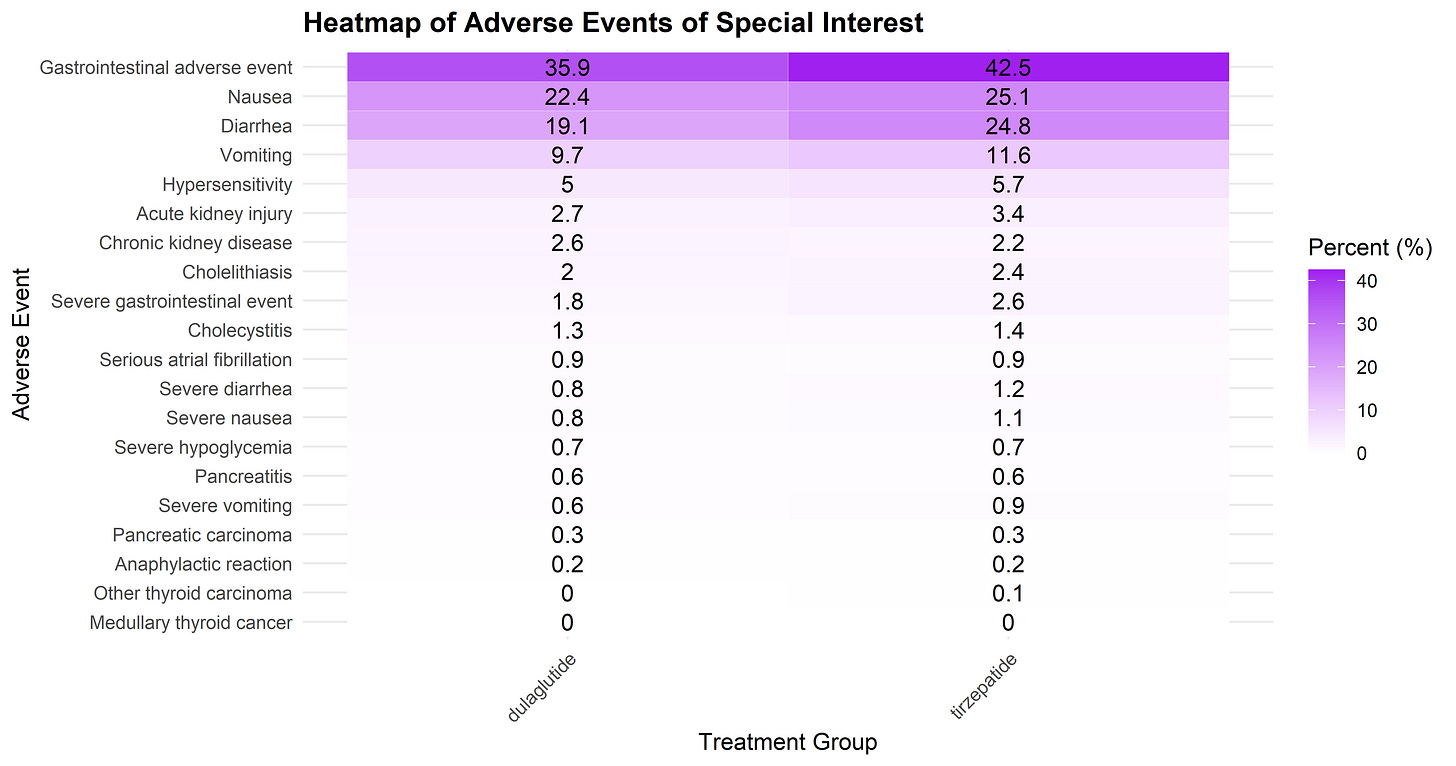
Gastrointestinal adverse events were more frequent in the tirzepatide (42.5%) compared with the dulaglutide (35.9%) group, although overall adverse‑event incidence between tirzepatide and dulaglutide was similar (89.6% vs 88.7%).
The main limitation of the trial was the use of a sub-maximal dose of the active comparator, dulaglutide (1.5 mg). Using the higher, more effective 4.5 mg dose of dulaglutide might have yielded different results regarding the gap in efficacy. The Moreover, the trial the use of an active comparator (dulaglutide) rather than a placebo, limits the interpretation of absolute treatment effects.
Time to dig into some more details.
The SURPASS program is a comprehensive Phase 3 global clinical trial program evaluating the efficacy and safety of tirzepatide (5 mg, 10 mg, and 15 mg sc once-weekly) in over 10,000 adults with type 2 diabetes. Studies consistently demonstrated that tirzepatide provided superior A1C reduction and body weight loss compared to placebo, semaglutide, and insulin. SURPASS‑CVOT extends to evaluating cardiovascular outcomes in patients with type 2 diabetes and established atherosclerotic cardiovascular disease.
Study design:
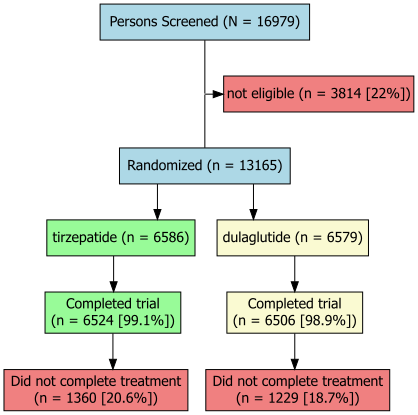
Trial type: Phase 3, Double-blind, active-controlled, randomized, parallel-group non-inferiority trial.
Randomization: 1:1 treatment allocation stratified by SGLT2 inhibitor use at baseline and country. Allocation concealed via interactive web response system.
Blinding: Double-blind (participant, investigator)
Population: Adults 40 years of age and older with a diagnosis of type 2 diabetes and established cardiovascular disease.
Setting: 640 sites in 30 countries
Treatment strategies: Maximum tolerated dose of weekly subcutaneous tirzepatide up to 15 mg vs dulaglutide 1.5 mg.
Primary endpoint: Time to first occurrence of a major adverse cardiovascular event (MACE).
Analysis methods: Time to event analysis
Summary method: Hazard ratio
Funding: Eli Lilly.
Trial Flow
Who was studied?
Trial participants needed to be at least 40 years of age, have a diagnosis of type 2 diabetes, HbA1c between 7% and 10.5%, BMI ≥ 25 kg/m2 and have established cardiovascular disease (coronary artery disease, cerebrovascular disease, or peripheral arterial disease).
Notable exclusions included type 1 diabetes, uncontrolled diabetes requiring an immediate intervention, any cardiovascular event in the 60 days prior to screening, having a known gastric emptying condition, history of proliferative retinopathy or macular edema, use of a GLP-1 receptor agonist or pramlitide in 3 months prior to first visit, contraindications to GLP-1 receptor agonist therapy. See supplemental appendix for more details on eligibility criteria.
At Randomization:
Average age ~ 64 years
~ 29% women
~ 65% had coronary artery disease
~ 19% had a history of a stroke
~ 25% had peripheral artery disease
~ 20% had a history of heart failure
Average duration of diabetes ~15 years
What were the treatment conditions?
Participants randomized to tirzepatide began at 2.5 mg weekly for 4 weeks, with 2.5 mg increases every 4 weeks through 5, 7.5, 10, 12.5, and 15 mg, until reaching the 15 mg target or their maximum tolerated maintenance dose. The full escalation period spanned 24 weeks (20 weeks of titration plus 4 weeks to steady state). Patients who initially tolerated only 5 mg or 10 mg underwent a second escalation at Visit 16 to attempt advancement toward 15 mg, recognizing that some participants better tolerated higher doses after prolonged exposure. Dose adjustments, including brief interruptions or single-step de‑escalation, were permitted for gastrointestinal adverse effects, with efforts focused on maintaining patients on treatment and resuming escalation when feasible.
Participants assigned to dulaglutide received 1.5 mg weekly from randomization and continued this dose throughout the trial. To preserve blinding during the tirzepatide titration phase, the dulaglutide group followed a sham escalation schedule with identical visit and dispensing frequency, though the active dose remained unchanged. If gastrointestinal tolerability issues prompted a “de‑escalation,” sites performed the same procedural steps used in the tirzepatide arm, but the actual administered dose remained 1.5 mg. After the escalation period, study drug was dispensed every 3 months, with all participants encouraged to remain on therapy and continue follow‑up regardless of temporary interruptions.
Main effects
Primary cardiovascular outcome
The primary composite outcome (cardiovascular death, nonfatal myocardial infarction, or nonfatal stroke) occurred in 12.2% of patients treated with tirzepatide and 13.1% of patients treated with dulaglutide, corresponding to a hazard ratio of 0.92 (95.3% CI 0.83–1.01). This met prespecified criteria for noninferiority (p=0.003) but did not demonstrate superiority (p=0.09).
Metabolic and secondary outcomes
Although not adjusted for multiplicity, tirzepatide produced greater improvements in several secondary outcomes, including:
Greater reductions in HbA1c and body weight
More favorable renal outcomes
A numerically lower rate of all‑cause mortality (approximately 16% relative reduction vs dulaglutide)
These findings reinforce tirzepatide’s robust metabolic effects while demonstrating preservation of established cardiovascular benefit when compared with a GLP‑1 receptor agonist with proven CV efficacy.
Safety
As expected, gastrointestinal adverse events (including nausea, vomiting, diarrhea, and constipation) were more common with tirzepatide, particularly during dose escalation.
Overall rates of serious adverse events were similar between groups, and no new safety signals were identified over long‑term follow‑up.
Limitations
The major limitations of this trial include:
The use of a sub-maximal dose of the active comparator, dulaglutide (1.5 mg). Using the higher, more effective 4.5 mg dose of dulaglutide might have yielded different results regarding the gap in efficacy.
An active‑comparator (non‑placebo) design, which precludes estimation of absolute cardiovascular benefit versus standard care.
No demonstration of cardiovascular superiority, despite favorable metabolic effects.
Limited population diversity, potentially affecting generalizability.
Secondary outcomes (mortality, renal benefits) were not controlled for multiple testing.
There were imbalances between the treatment groups in the proportion of patients starting a SGLT2 inhibitor post-randomization.
Even with its limitations, SURPASS‑CVOT provides clear evidence that tirzepatide delivers cardiovascular safety and matches the performance of a proven GLP‑1 receptor agonist in a high‑risk secondary‑prevention setting.
In context
Compared with earlier cardiovascular outcome trials, SURPASS‑CVOT aligns more closely with REWIND than with LEADER, SUSTAIN‑6, or SELECT in both design and inference. LEADER demonstrated clear cardiovascular superiority of liraglutide versus placebo in high‑risk patients with type 2 diabetes, while SUSTAIN‑6 similarly showed significant MACE reduction with semaglutide in a predominantly high‑risk diabetic population. In contrast, REWIND showed a modest but significant MACE reduction with dulaglutide in a broader, earlier‑risk diabetes population. In contrast, SURPASS‑CVOT tested noninferiority, showing that tirzepatide preserved, rather than exceeded, the established cardiovascular benefit of dulaglutide when used in patients with diabetes and established ASCVD, while delivering greater metabolic effects. SELECT differs fundamentally as it demonstrated cardiovascular superiority of semaglutide versus placebo in people with obesity without diabetes, establishing weight loss itself as a cardiovascular intervention. Taken together, LEADER and SUSTAIN‑6 demonstrate robust cardiovascular risk reduction for daily liraglutide and weekly semaglutide in predominantly high‑risk patients with type 2 diabetes, while REWIND extends this benefit to a broader, lower‑risk diabetic population. SELECT extends benefit to non‑diabetic obesity, and SURPASS‑CVOT reassures clinicians that tirzepatide achieves cardiovascular safety and equivalence to proven GLP‑1 therapy rather than incremental CV risk reduction.